Trace elements perform important physiological functions, such as the complex functions in metabolism, and deficiencies can lead to general disorders, which is why they are provided in the feed. Though important for maintaining optimal livestock health and performance, the importance of benefits from trace element supplementation tend to be underestimated.
Trace element absorption challenges
Trace element bioavailability, or the degree to which or rate at which it is absorbed after administration, varies based on a number of circumstances. For example, the reasons for variations are influenced by the different digestive tract anatomies between the species, the age of the animal (intestinal uptake capacity decreases with age), animal health and the presence of stress. In addition, trace element bioavailability depends further on a number of factors, such as: the status of the trace element; its ability to form chemical bonds; the trace element's dosage; and the antagonists which inhibit absorption. These antagonists require highly bioavailable trace elements to guarantee a sufficient supply to the animal even at a low feed concentration.
Unbalanced or high levels of certain trace elements lead to shortages of the other elements. For example, a zinc surplus will cause copper transport across the intestinal epithelial cells, or enterocytes, to be blocked due to the formation of intestinal metallothionein. This is also recognized as a secondary copper deficiency.
Beyond these, mycotoxins, crude fiber, tannins and phytic acid affecting the availability of trace elements.
Characterization, differentiation of trace elements
The various feed additives approved as trace element sources can be distinguished in terms of their availability for the animal. The best known and largely used inorganic trace minerals, sulphates and oxides, vary mainly due to their solubility in the aqueous medium of the digestive tract. A product with very low solubility, e.g. copper oxide, should therefore be avoided in livestock feed.
However, the high solubility of sulphates provides the disadvantage of very reactive copper ions, i.e. a strong pro-oxidant initiates and hurries oxidation, which promotes adverse reactions with feed components. This causes the degradation and damage of sensitive ingredients, such as vitamins or fat, as well as forming of harmful free radicals and peroxides and reducing palatability of feed.
In contrast to inorganic, organically bound trace elements, or chelates, are stable in the diet and improve absorption due to their chemical structure. The advantage is superior in isolation from the solubility of inorganic trace elements, but also in reducing absorption-inhibitory responses in the digestive tract. The positive effects of organically bound versus inorganic trace elements have been confirmed in numerous scientific studies.
Nevertheless, an adequate assessment of the quality of organically bound trace elements can be easily checked by simple analytical methods.
About hydroxyl trace minerals
A new category of trace minerals, known as hydroxy trace minerals, belong to the group of inorganic trace mineral sources, but have similar chemically stable structures such as organically bound trace elements. Covalent bonds and a unique crystalline matrix structure provide a reliable stability, which differentiates hydroxy trace minerals from both organic and inorganic minerals.
In contrast to copper sulphate, these products are hardly soluble at neutral pH, which provides increased trace mineral stability in the intestine. Less-reactive trace element ions occur in the upper digestive tract and are associated with fewer interactions with other feed components. The crystalline structure releases the trace elements slowly over the full length of the small intestine resulting in improved trace element absorption and enhanced effects on intestinal health.
Effects of various trace element sources in feed
Trials have demonstrated improved stability of sensitive feed ingredients through stable trace element sources, such as chelates or hydroxy trace minerals in comparison to sulphates. Such results are based on the non-hygroscopic and non-reactive (non-pro-oxidative) characteristics of these trace element sources.
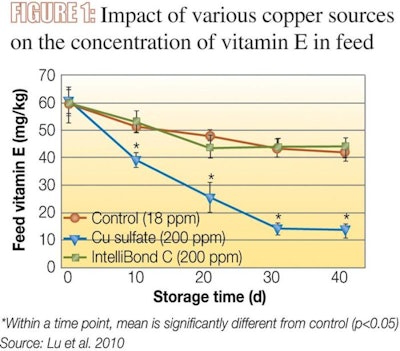
For example, copper from copper sulphate at a high dosage - as opposed to hydroxy copper - presents a very strong negative impact on vitamin E stability in feed (Figure 1). Due to high solubility and high reactivity (i.e. formation of insoluble complexes with phytate), increased dosages of copper sulphate result in a significant negative impact on phytate phosphorus hydrolysis. Both copper from copper lysine and hydroxy copper did not influence phosphorus hydrolysis (Figure 2).
T he usable amount of trace elements is crucial to the animal. This requires accurate knowledge of the composition of the feed and, if possible, the diminishing availability of trace elements and interactions with other feed ingredients. The trace element supply can be improved by the use of high-quality trace mineral sources such as organic chelates, however, the assessment of the used quality is essential.
T he positive effects of chelates and hydroxy trace elements in feed have been proven to increase the stability of sensitive feed components and ingredients and produce higher bioavailability.

















