This article appears in the September issue of Pig International. View all of the articles in the digital edition of this magazine.
Pig production has responded to the challenge of increased global demand for pork by a process of consolidation and increased size to capture the benefits of economies of scale. Over the last 25 years, the swine industry has evolved in order to increase production performance, health and animal well-being towards age-segregated or multiple site production models.
In general, it can be stated that the changes in production systems have produced a positive effect in the health status of pigs by improving the way we raise pigs today. However, bigger and more complex farms and production systems require that we approach disease control in a different way by recognizing the impact throughout the production cycle or production chain. The prevention chain approach, outlined here, applies new and existing knowledge in swine diseases management.
Infection and prevention chain thinking
The infection/prevention chain concept brings together new and existing knowledge in swine diseases management, connecting epidemiological events (infection chain) between the different production phases (production chain) using logical chain-thinking to create multi-phase intervention strategies (prevention chain). This systematic approach to disease control was first proposed by Dr. Eduardo Fano, Dr. Brian Payne and Dr. Edgar Diaz in the Unites States to address both M. hyopneumoniae and porcine reproductive and respiratory syndrome (PRRS) control, but it can be applied to practically all infectious diseases.
Bigger and more complex farms and production systems require that we approach disease control in a different way.
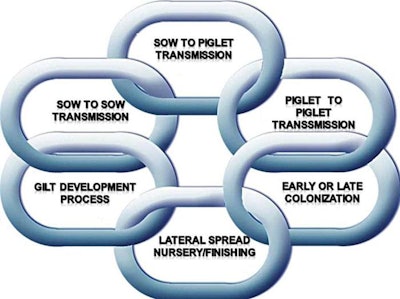
Persistence of infection, shedding patterns, vertical transmission, horizontal transmission and lateral infection should be considered as critical epidemiological pieces participating in the “infection chain” of swine pathogens throughout the complete production process (whole herd), resulting in the clear manifestation of clinical symptoms with the correspondent production and financial losses due to increased mortality, culling rate, treatments and lack of performance, even in subclinical disease. This “chain” starts with gilt development and introduction into the sow herd and ends in growth-finish populations or re-connects with a new link with gilt production.

Logical chain-thinking is used to create multi-phase intervention strategies.
Part of this thinking relies on the simple concept of an infection or disease management equation: to successfully control a disease, we need to minimize exposure (reduce infection) and maximize immunity (increase protection).

To successfully control a disease, we need to minimize exposure (reduce infection) and maximize immunity (increase protection).
This “chain thinking” allows a more holistic and comprehensive approach to understand the epidemiology and prevention of major infectious agents by matching "production chain” with “infection chain,” resulting in a “prevention chain” extremely focused on the entire production system at all stages instead of just the individual piglet. Therefore, this approach can help to modify or re-adjust the intervention strategies (pig flow, internal biosecurity management and prevention protocols) applied for control, prevention and even elimination of disease, considering the whole infection chain of the agent, allowing an appropriate prevention chain approach. The goal is to create a way of thinking where understanding the root of the problem is the first step of a whole herd approach to health management.
Lessons learned from the prevention chain approach
After years of failed interventions, the implementation of a systematic prevention chain approach based on improved diagnostic tools, better understanding of the epidemiology of PRRS and benefits of biosecurity has allowed successful PRRS control, e.g., using load-close-homogenize protocols.
The first step is to bring in as many gilts as are available and can be housed into the herd (load). Next, all breeding age animals in the herd are immunized twice 30 days apart with MLV PRRS vaccine (homogenize), and then no new animal introductions are allowed until the breeding herd has been declared stable (close). A breeding herd is classified PRRS-stable-positive when previously exposed sows are producing PRRS-negative piglets at weaning and no clinical signs consistent with PRRS are observed in breeding herd.
By systematically applying these methods and measuring the outcome you can establish two novel metrics: time to PRRS stability (time to negative), and time to return to baseline production (when breeding herd output returns to pre-outbreak levels). Using time to stability to measure success, it was discovered (again, using chain thinking) that cross-fostering practices in the farrowing house extended the herd closure time on a number of herds because the movement of pigs maintained PRRS virus circulation for a longer time. Once a farm is weaning PRRS-negative piglets, the prevention chain approach focuses on the next critical control point; is there a need to protect piglets against PRRS? Are these pigs going to highly dense grow-out sites? If these pigs break, will they increase the risk of infecting the neighbor herds? How much money am I leaving on the table by not protecting them? The systematic, whole-herd approach (gilts, sows and piglets) allows you to make better disease control decisions.
Applying the prevention chain approach to control M. hyopneumoniae (Mhp) infections in production systems has led to an understanding of the importance of the gilt in the control of this costly disease. Once a growing gilt is infected she will remain a carrier for over 200 days; bringing a large number of replacement gilts into a herd, like during a herd expansion, could lead to increased Mhp circulation in the breeding population, higher rates of vertical transmission to the piglets and more piglets infected at the time of weaning. This increase could lead to an imbalance in the infection pressure equation towards more disease. By applying specific disease management of the gilts with vaccination, short and strategic antibiotic treatments or sufficient acclimatization time, we can reduce the likelihood of this event.

Applying the prevention chain approach to control M. hyopneumoniae (Mhp) infections in production systems has led to an understanding of the importance of the gilt.
Take home message
The infection/prevention chain concept applies new and existing knowledge in swine diseases management:
- It encourages systematic interventions: The infection/prevention chain concept is a tool for swine diseases management, using the whole-herd approach.
- It connects epidemiological events (infection chain) between the different production phases (production chain) and facilitates the root cause analysis.
- It utilizes a logical chain-thinking to create multi-phase intervention strategies (prevention chain), critical to achieve successful and sustainable health programs.
Read more: Health is the biggest barrier for rearing gilts in the US

















