Salmonella infection
The oral route is the natural pathway of infection of chickens with Salmonella enteritidis. Following ingestion, the organisms invade enterocytes lining the intestinal tract and then pass to the underlying mucosal associated lymphoid tissue (MALT). SE bacteria enter the blood system, probably when phagocytosed (absorbed) by leucocytes, eventually reaching the spleen, liver and bone marrow. Intestinal replication in internal organs and excretion of viable SE through the feces will result in extensive environmental contamination.
Infection of newly hatched chicks results in a massive multiplication of SE, with large numbers being excreted in the feces for several weeks. In contrast, older birds are less susceptible to infection. This age-related resistance has been attributed to the development and maintenance of a stable microflora, which competes with salmonella for carbon sources and receptors on the enterocytes. In addition, established microflora may secrete metabolic products which inhibit the growth of salmonellas, including SE.
Salmonella vaccines
Several research studies have shown the benefits of using live attenuated and inactivated vaccines alone or in combination to prevent SE in flocks. Benefits of vaccinating against SE include a reduction in organ invasion and fecal shedding (lower risk of environmental contamination) and lower levels of both horizontal and vertical transmission (lower risk of egg contamination).
SE inactivated vaccines are characterized by their high antigenic concentration and capacity to stimulate the production of specific antibodies (humoral immunity) in the hen. Vertically transmitted antibodies (Immunoglobulin G) are found at high concentrations in the yolk, providing some protection against internal SE contamination. Subcutaneous administration of oil emulsion inactivated vaccines to laying hens has been reported to significantly reduce the frequency of SE isolation from internal organs and decrease fecal shedding.
Live attenuated Salmonella typhimurium vaccines are associated with a more diverse protective immune response in poultry, when compared with inactivated vaccines.
Live attenuated vaccines provide protection against mucosal and systemic infection and indirectly reduce intestinal, environmental and egg contamination. Live attenuated vaccines are characterized by:
- Stable attenuation. This is achieved by modifying genes coding for metabolic and virulence functions, while preserving the expression of key determinants required for immunogenicity against SE. Double or even triple mutations in genes such as Cya/Crp (regulation of carbon source utilization). Aro A (synthesis of purine or amino acids) or Gal E (biosynthesis of LPS), effectively reduce the risk of reversion to pathogenicity. As a result of attenuation, vaccine strains are characterized by their prolonged generation time, allowing the development of a specific immune response with the eventual elimination of the administered vaccine strain by the immune system.
- Safety for non-target species and humans. This is self evident but is subjected to intensive testing for safety.
- Efficacy. When administered to newly hatched chicks, live attenuated vaccines multiply extensively due to the absence of the complex normal microbial flora found in chickens older than 4 weeks of age and older birds with a fully functional immune system. The early multiplication of the normal flora prevents colonization by field strains of salmonella. The practical consequence is that administration of live vaccines to very young chickens by drinking water or coarse spray provides protection within hours against invasion by salmonella. This early protection, achieved through colonization-inhibition, is followed by the development of a normal immunity within few weeks.
- Capability for Mass administration by coarse spray and in drinking water. When administered at an early age, ST vaccine strains are able to effectively infect and colonize the gastrointestinal tract, denying field SE and non-Group D Salmonella strains access to receptor sites on intestinal cells.
Designing a vaccination program
Vaccination programs should be developed on the basis of the risks and consequences of infection, especially following the introduction of the FDA “Final Rule.” The previous history of SE contamination in pullets and production units and the level and implementation of biosecurity and beneficial management practices are significant considerations in developing an effective program. Live vaccines stimulate cell-mediated immunity (CMI), leading to clearance of salmonella from the internal organs and a reduction of salmonella shedding into the environment. Inactivated vaccines stimulate the development of antibodies, which provide protection not only to the hens during production but also to the internal contents of the egg.
Vaccination programs based only on live attenuated vaccines should include three doses. The first dose is usually administered at the hatchery or during the first two days of age after delivery to the rearing unit by coarse spray or drinking water. Vaccination at the hatchery allows for more uniform administration, efficient infection and colonization of the gastrointestinal tract by vaccine strains and development of early protection against field strains of salmonella by competitive exclusion. The second and third doses can be administered between 4-6 weeks and 14-16 weeks of age respectively by coarse spray or drinking water. The use of live vaccines with a high antigenic concentration is recommended for the 2nd and 3rd vaccinations at the rearing farm due to the presence of a stable microflora which may compete with the vaccine strains.
A live vaccination program can be reinforced by the administration of at least one inactivated vaccine, to stimulate the development of antibodies to protect against colonization and excretion of SE in the environment and vertical transmission to eggs.
Combined programs can be designed which incorporate two (1 day and 4-6 weeks) or three (1d, 4-6 weeks and 16 weeks) live vaccines with an additional inactivated vaccine at around 12 weeks. Under very high risk of SE contamination, including placements in units shown previously to be environmentally positive, the use of two live (1 day and 4-6 weeks) and two inactivated vaccines (10 and 14 weeks) should be considered.
In commercial layer flocks undergoing molting, live vaccines may be administered according to the directions of a poultry health professional under an off-label protocol prior to feed withdrawal. This approach is recommended to prevent the known exacerbation of SE problems in flocks under severe stress attributable to molting, during which the potential for contamination may exist.
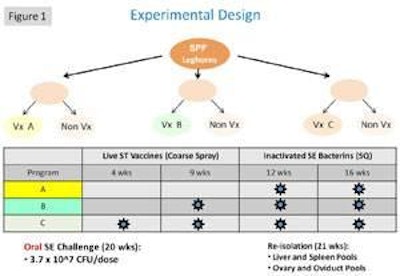
Vaccination programs vs. protection against challenge with S.E. and S. Heidelberg
Internal research studies conducted by Lohmann Animal Health International have been completed to evaluate the level of protection provided by different vaccination programs against challenge with SE and Salmonella heidelberg strains. Those studies evaluated the level of protection provided by different vaccination programs based on inactivated or live and inactivated vaccines against colonization of internal organs, ovaries and oviduct.
The studies (Figure 1) show the benefits of vaccination against SE. A significant level of protection against organ (spleen and liver pools and ovary and oviduct pools respectively) was obtained following challenge with a high dose of SE in vaccinated hens irrespective of the vaccination program used (Figures 2 and 3). The highest reduction in SE positive tissues was observed in birds vaccinated with both, live and inactivated vaccines (Figure 4). This finding confirms the development of a solid immune response against SE, achieved by the stimulation of both, local (gastrointestinal tract) and cell mediated immunity by live attenuated vaccines and humoral immunity (antibodies) attributed to highly immunogenic inactivated vaccines.
Adapted from a presentation made at the 2011 Midwest Poultry Federation Convention.

















