A disease causing significant lameness and mortality problems in broilers flocks has emerged as a problem in several U.S. broiler producing areas in the last year and a half. Increased mortality is seen after four weeks of age and can reach as high as 10% to 15%, thus resulting in very poor performance results.
The disease was first mistakenly named “kinky back,” which is a descriptive term for this emerging disease problem now seen in several broiler producing areas since early 2008. The actual lesion is an abscess in the free thoracic vertebrae that produces paralysis of the bird due to pressure placed on the spinal cord in that area. The abscess is associated with the isolation of the bacteria Enterococcus cecorum.
Widespread presence in U.S.
This disease was first seen in young, growing broiler breeder males in North Carolina in 2006. The broiler problem was first seen in Pennsylvania in January 2008. Since then, the disease of broilers has been reported in Washington, North Carolina, South Carolina, Arkansas, Mississippi, Alabama and California. Additionally, broilers in Canada, the Netherlands and Belgium have been reported to show lameness from spinal abscesses. In Pennsylvania, the problem is widespread and has been seen in flocks of all five of the state’s broiler companies.
This problem is not seen in all flocks but only sporadically. In one broiler complex, for example, an average of two houses of twenty placed in a week showed spinal abscesses. The problem does not appear to spread from an affected house to another during growing. Only certain houses on a farm will be affected. Once a house has affected birds, subsequent flocks will be affected, and the causative agent appears to persist in the environment.
Culling in flocks
Males are predominantly affected. All broiler strains have been affected. The effect on the flock is due to culling of affected birds as they become lame and unable to get to feed and water. Initially, the percent of affected birds in a flock was relatively high at 5% to 10% percent. Now, the percentage of birds affected is lower, typically 2% to 4%.
The typical history is for a flock 30 days or older showing birds sitting on their backsides in a very typical stance. Some will also lie on their sides, paralyzed. Affected flocks have been as young as 15 days of age. In the worst cases, 50 to 200 birds need to be culled each day until marketed.
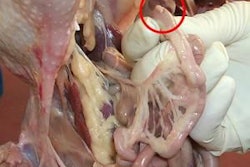
Affected birds that are necropsied will not show the typical lesions associated with lameness such as femoral head necrosis or synovitis. The seventh thoracic vertebra, the free thoracic vertebra, contains the abscess and shows a palpable swelling. This vertebra is located just caudal to the last rib and beside the cranial lobe of the kidney. The abscess is visualized by cutting through the vertebra with a heavy, sharp scissors or knife at a right angle to the vertebra. The cross section will show an abscess in the vertebral body. If the vertebrae are cut longitudinally, one can see the compression placed on the spinal cord resulting in paralysis.
This disease needs to be differentiated from true ‘kinky back’ (spondylolisthesis) and scoliosis. True kinky back is a genetic condition resulting in anterior-posterior rotation of the bodies of the free thoracic vertebrae resulting in pinching of the spinal cord just anterior to the kidney. Scoliosis is another genetically associated disease resulting in lateral curvature of the thoracic vertebrae causing paralysis.
Caused by gram-positive bacterium
The causative organism of this disease appears to be Enterococcus cecorum, a Gram-positive bacterium. Initially, the method used to identify bacteria in our laboratory, the Sensititer ARIS system, reported the bacterium to be Streptococcus suis. Further tests, using the MicroSEQ system, were used to sequence the DNA and compare it to a DNA databank in order to identify the bacterium as Enterococcus cecorum. This bacterium is a normal inhabitant of the intestinal tract. It also has been reported to cause a septicemia and synovitis in broiler chickens. Typically, E. cecorum is sensitive to gentamycin and penicillin but resistant to tetracyclines, sulfas and streptomycin.
The source of the infection is not known. It is believed that the breeder hatching eggs may be a possible source. One search for the bacterium in trays of hatching chicks by swabbing yielded E. cecorum in one of six breeder sources assayed. Theoretically, hatch egg shells or shell membranes contaminated with E. cecorum could contaminate the entire hatcher cabinet. Another theory is that E. cecorum becomes blood-borne from the intestine. In human infants, vertebral abscesses are common sequelae to navel infections (hematogenous spread from omphalophlebitis). Histologic examinations of the cecae of affected birds show evidence of an adherent and effacing strain of E. coli which is theoretically allowing the Enterococcus cecorum bacterium entry into the blood.
Whatever the source of the infection, once E. cecorum is blood-borne, it invades the vertebral body of the free thoracic vertebrae resulting in necrosis, inflammation and swelling of the vertebral body. The resultant swelling causes compression on the spinal cord and/or pathological fractures of the vertebral body.
Prevention and treatment
Treatment has been to cull the affected birds, sanitize water, and give as good an environment as possible. Antibiotics have not been used as the potentially useful antibiotics are not approved in the U.S. for use in broilers at that age.
Preventative antibiotics such as tylosin and amoxicillin have been used in Europe during the grow-out to stop the recurrence of the disease. Also, increasing the dosage of gentamycin used in the Marek’s vaccine from a half dose to a full dose has been tried.
Preventative measures after the first flock where the disease has occurred that has shown success are as follows:
- Complete cleanout and disinfection
- Fumigation of the house
- Water line removal of biofilms and bacteria
- New litter
- Continuous water line sanitation during grow-out
Persistent management needed
Elimination of the disease usually does not occur after only one round of cleaning and disinfection. Increased efforts in subsequent flocks have been needed to eventually eliminate the disease. These measures are as follows:
- Compost litter between flocks
- Fog disinfectant or fumigate
- Water line biofilm and bacteria removal
- Continuous water line sanitation
Composting of litter between flocks is becoming a more routine practice in the broiler industry to reduce litter pathogens and darkling beetles. Litter composting utilizes both caked and normal litter. Special equipment pulverizes the litter and places it into two or three piles the length of the house. After three days, if the litter has 25% to 30% moisture, the pile will heat to 130 F or more. The piles are turned and allowed to reheat to 130 F or more. Multiple turnings are ideal. At least 10 days downtime is required for a satisfactory composting of litter.
Answers in future research?
Future research will hopefully answer questions about E. cecorum and vertebral abscesses, including the following:
- Better identify the source(s) of the causative bacteria
- Determine if intestinal microflora imbalance is involved with the disease
- Improve the effectiveness of preventative measures
- Develop effective treatment(s) for affected flocks to minimize losses
Until more is known about the source and cause of this emerging disease syndrome, producers should practice culling, proactive husbandry and cleaning and disinfection in affected flocks and facilities.