Porcine epidemic diarrhea virus (PEDV) is a highly contagious disease characterized by vomiting, diarrhea and inappetence in pigs of all ages. Mortality in young piglets is generally less common than in transmissible gastroenteritis (TGE).
PEDV first diagnosed
First described in Britain in 1972, PEDV has been identified in most European countries, China, Korea, Japan, India, Indonesia, Taiwan and now in North America. However, the PEDV antibody was absent from sera from small numbers of pigs from Sweden, Australia, Northern Ireland, and as of early 2013 from the United States. Similar diseases not confirmed as epidemic diarrhea have been identified in Canada. In Austria and Switzerland serological surveys suggest that the incidence is low in contrast to Belgium.
Aetiology
The causal agent is a coronavirus distinct from TGE, PRCV and P.H.E.V. The virus is 95-150 nm (mean 130 nm) in negative stain and 60 nm in section. The peplomeres are 18-23 nm in length. There are three major protein antigens, the S or spike glycosylated protein of 85-130 kDa, the M protein of 27 kDa and an N protein of 58 kDa with some relationship to Feline Infectious Peritonitis virus.
All isolates appear to belong to a single serotype. It can be cultivated in Vero cells with added trypsin and produces syncytia on subculture and has been sequenced. Sequence comparisons show that Chinese and Korean isolates form clusters distinct from European isolates and that recent Chinese isolates differ from Korean strains. The virus is destroyed at >60oC within 30 minutes, but is stable at 50oC. At 37oC it is stable between pH 6.5 and 7.5 but at 4oC only between pH 5.0-pH 9.0.
Pathogenesis
The PEDV virus does not replicate in the respiratory tract but otherwise pathogenesis resembles that of TGE. It attaches to a 150 kb receptor on epithelial cells, and appears to require a greater density of porcine aminopeptidase receptors in cell lines that TGE. Epithelial cells at the tips and sides of the villi are infected and can shed virus without being destroyed, but, by the onset of diarrhea, epithelial cell shedding has already begun.
Six hours later it occurs in the ileum. Levels of disaccharidases, alkaline and acid phosphatase, succinic dehydrogenase and monoamine oxidase fall. In older pigs lactase levels have also been shown to fall. Villous fusion and atrophy are widespread within 24 hours of the onset of clinical signs. In partially-immune piglets, a much localized area of the small intestine may show villous atrophy. Serum antibody levels become detectable 2-4 weeks after infection and rise to levels of up to 1:10,000 declining rapidly to lower levels of 1:20-1:640.
Passive antibody has disappeared from the sera of piglets within 5-13 weeks and active immunity then develops. The presence of serum antibody in a nursing sow does not rule out epidemic diarrhea in her litter. Virus shedding in the feces continues for at least 8 days. Antibody levels on infected farms peak at 4 months and decline from then onward.
Clinical PEDV signs
PEDV occurs as an explosive outbreak of diarrhea in non-immune weaned pigs or in pigs of all ages. After an incubation period of 1-3 days, piglets develop a TGE-like watery diarrhea and may vomit. Although morbidity in non-immune pigs approaches 100 percent, mortality can be low. The disease spreads rapidly in the unit. Twenty to 80 percent of the older pigs may be affected with vomiting and diarrhea. One hundred percent inappetence may occur, especially in breeding stock.
Clinical signs can be restricted to older animals with sucking and young weaned pigs under 32 kg live weight are rarely affected. Affected weaned pigs are dull and unwilling to rise. Pyrexia is rare. The diarrhea is greenish-brown in color and very fluid, often coating the pigs completely. Signs of dehydration are common. Vomiting is prominent in some outbreaks and less obvious in others. The acute stage of vomiting and diarrhea lasts approximately 3 days but recovery takes a further 7-8 days.
Mortality is rare in the absence of intercurrent disease and where water is freely available. Affected feeding pigs take about 14 days longer to reach 90 Kg due to the effects of inappetence and loss of condition. Clinically-recovered pigs show no after effect.
Agalactia may occur in sows and result in some indirect mortality in unaffected litters, and productivity in subsequent litters may be reduced, particularly in gilts. Severely dehydrated, wasted piglets not responding to supportive therapy should be humanely destroyed.
Pathological findings
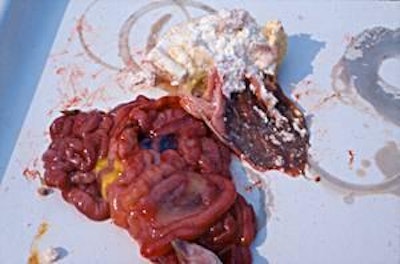
Few weaned pigs have been examined as mortality is low. The stomach is usually empty or filled with bile-stained fluid and both large and small intestines are pale and thin-walled, often with fluid contents. Villous atrophy is present. Some villous atrophy may be seen in the small intestines of baby pigs affected by this condition and they may have gastritis.
Epidemiology
Separated in 1971 from TGE on epidemiological grounds in the 1969-1977 UK TGE outbreak, the disease continued until spring 1972 and then died down in the summer to reappear in winter 1972-73. Pig contact is important and spread has occurred through pig movement, markets and by transport trucks.
Disease in weaned pigs was first described in 1977. Epidemic diarrhea is usually introduced by carrier pigs and introduction may be followed by an explosive outbreak. Severe outbreaks involving sows and piglets are now unusual in the UK and clinical disease may be confined to individual litters or occur in recently purchased grower pigs. Outbreaks have occurred in Italy (2005-2006), China, Korea, Japan, and currently in the US.
PEDV diagnosis
PEDV may be suspected in weaned animals by its history and clinical signs of an acute, rapidly spreading scour with vomiting and inappetence not affecting sucking pigs. The only similar condition is TGE, which can be eliminated by laboratory tests. When it affects all ages, it may be suspected by the pattern of mortality in a non-immune herd.
In a partially immune herd, only a few piglets may develop the disease and differentiation from E.coli
(absence of agent, villous atrophy) clostridia, cryptosporidia and coccidia (no blood or necrosis of the small intestinal epithelium) and rotavirus and TGE (laboratory methods including immunofluorescence, RT-PCR and ELISA must be carried out). Confirmation can be obtained by immunoelectron microscopy of the intestinal contents or feces of affected pigs when coronavirus particles may be seen.
The virus may be demonstrated using ELISAS, RT-PCR, nested RT-PCR and quantitative RT-PCR. Specific fluorescent antibody or immunoperoxidase demonstrates the virus in intestinal epithelium and an ELISA test is available for the detection of specific antibody in the sera or milk of recovered pigs. Only rising titres in paired serum samples are diagnostic. A number of ELISAs have been evaluated. The most efficient appears to be one using whole, tissue culture virus as antigen although the S protein can also be used. It has become clear that dual infections of TGE and PEDV can occur in the same pig.
PEDV treatment, control
Provide adequate water and provide milk substitute for any piglets of affected sows which may lose their milk and adequate water for all affected stock at all times. Piglets may be treated using glucose, such as glycine electrolyte solutions.
There is no specific treatment, although pig interferon has been used to reduce weight loss and mixed monoclonal antibodies and egg yolk antibody have also been found to protect. The effects of an outbreak of PEDV may be controlled by isolating all sows within 14 days of farrowing, and by infecting all those due to farrow in more than 14 days.
Weaners also may be infected to reduce the duration of the outbreak. Where swine dysentery or other intercurrent disease is present, recovery from PEDV is hastened by the treatment appropriate to the intercurrent disease. Vaccines attenuated by passage (83 times) have been described and protect 94.6 percent of pigs after 21 days, but are not available in Europe. Genetically-engineered plant protein and Lactobacillus vaccines have been produced. For more information, see www.pigdiseases.co.uk.

















